Anyone who’s ever heard of a dinosaur almost certainly also knows about the pterosaurs- the flying reptiles that often serve as window dressing or to give a sense of scale to depictions of dinosaurs in prehistoric artwork. They also, to the eternal exasperation of paleontologists, get labeled by the media as ‘flying dinosaurs’. But the pterosaurs deserve better, and the study of their diversity and biology makes for a fascinating look into the Mesozoic Era.
To begin with, it bears repeating that pterosaurs are not dinosaurs, as they don’t have the anatomical features necessary for this classification (see our article on what it takes to be a dinosaur for more on this). But they’re close to dinosaurs, on the same side of the archosaur family tree, and are probably the closest major group to the dinosaurs themselves. They also showed up and died out at around the same time as dinosaurs too, arising in the mid-late Triassic and going out with a bang at the end of the Cretaceous.
Many people casually refer to pterosaurs as ‘pterodactyls’, a practice that has actually been going on for centuries, but is also technically incorrect. ‘Pterodactyl’ really just refers to members of the genus Pterodactylus, and the group as a whole is properly known as the Pterosauria (‘winged reptiles’). Pterodactylus itself is actually a fairly small member of this group, about the size of a duck when fully grown, but it has the distinction of being one of the first pterosaurs ever discovered, and predates the first scientific description of any dinosaur. Wonderfully preserved examples of this animal can be found in the Solnhofen limestone near other paleontological landmark Archaeopteryx.
Pterosaurs have the distinction of being the first flying vertebrates in history, beating birds (the true flying dinosaurs) and bats to the punch. This ability required special anatomy, and the bodies of pterosaurs were marvels of evolution. Like bats, their wings for formed out of skin membranes supported by the arm and hand. However, the membranes of bats are spread across all the digits, while the wings of pterosaurs are supported by a single, extremely long 4th or ‘ring’ finger. The wing membranes were not simple flaps of skin, but were packed with some sort of tough material called actinofibrils. The exact composition of these structures is still uncertain, but they may have helped strengthen the wing membrane and maintaining its rigid yet flexible structure (Bennett, 2000). Like birds, pterosaurs had hollow bones, a respiratory system with a series of air sacs throughout the body, an enlarged brain, and a keeled sternum for anchoring large pectoral muscles. All of these are important adaptations for flight, meaning pterosaurs were likely quite sophisticated in the air.
Despite this commitment to flight, pterosaurs were not helpless on the land (Unwin, 1997). The shape of their skeletons and evidence from trackways tells us that pterosaurs competently walked on all four limbs when on the ground, using their first three fingers on their massive forelimbs for support, while the wing digit was held aloft. They walked on their entire back foot just as humans do, with the heel touching the ground (a condition known as being plantigrade), unlike dinosaurs who walked on only their toes. Some pterosaurs were also likely competent swimmers, and some species which lived over water may have habitually swam or dove.
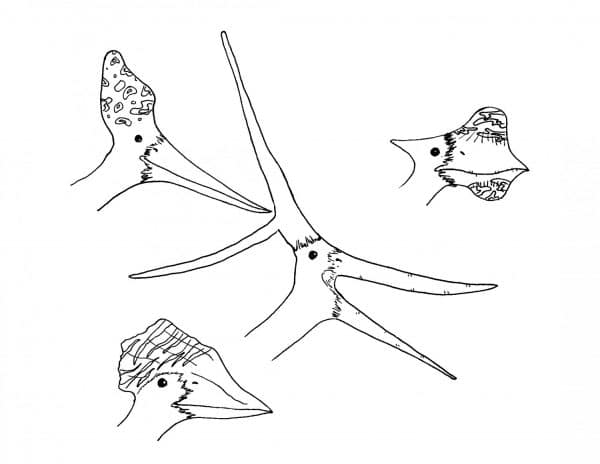
Many species of pterosaur had crests of some shape or another. Many crests were small and made up of mostly soft tissue, but some groups, like some pteranodonts and tapejarids, had massive bony crests with elaborate shapes. These crests were almost certainly for display, and could very well have been brightly coloured in life. Displaying pterosaurs, perhaps even colonies of them, would have been an amazing sight of the Mesozoic world.
Strangely enough, despite the fact that pterosaurs were not birds, and flew on membranous wings, they might have had feathers of some form. For decades, pterosaurs have been known from well-preserved specimens to have been covered in a fine coat of hair-like structures called pycnofibers. While not related to the hair of mammals, paleontologists suspected that pycnofibers could have been used for maintaining the body temperature of pterosaurs. This would indicate that they were warm-blooded, which would make sense for animals that led active, energy-demanding lifestyles. It has been suggested in the past that pycnofibers might be homologous to feathers, as they’re similar in structure and composition to primitive feathers seen in many dinosaurs. This idea was backed up in 2019 by a study describing several different pycnofiber structures on pterosaurs from Asia, some of which were extremely feather-like (Benton et al., 2019). This led some paleontologists to suggest that simple, hair-like feathers were present in the ancestor of both pterosaurs and dinosaurs, further developing or being lost in certain lineages. However, with the gaps in our knowledge of feather presence or absence in certain dinosaurs, this notion is still not fully accepted.
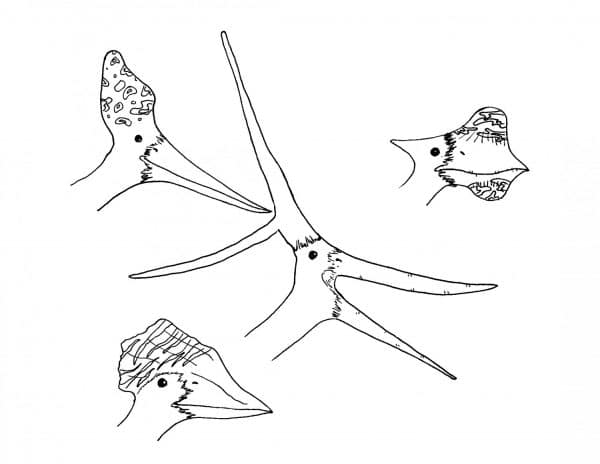
Some of the crazier pterosaur crests known to science. Perhaps not aerodynamic, but they helped these species pass on their genes. By Nicholas Carter
The ecology and evolution of pterosaurs is quite varied for a group of animals normally relegated to flying, prehistoric monsters in the mind of the public. The exact ancestry of pterosaurs is actually still quite mysterious. The earliest known pterosaurs are already adapted for flight, and unlike birds, we don’t have any of those handy transitional fossils between non-flying and flying species yet. One animal that often get pointed to as an early pterosaur relative is the small Triassic creature Scleromochlus, however, this animal itself is poorly understood (Benton, 1999). The highly generalized trend in pterosaur evolution is that earlier groups from the Triassic and Jurassic were mostly small, robin to gull-sized animals with toothy snouts and long, rudder-like tails. More derived groups from the late Jurassic to the end of the Cretaceous led to many strange forms, some of which developed those unusual crests, toothless beaks, and some extraordinarily huge species.
Some pterosaurs may have lived lifestyles similar to small or medium-sized birds and bats today. Insects had already evolved flight long before pterosaurs, and were a highly available food resource in the Triassic. Early pterosaurs often get characterized as small, insectivorous animals who used their short wings and long tails to swoop and turn with great agility after their prey. Some, like meter-long Dimorphodon from the early Jurassic of England, may have preyed on small vertebrate animals along the prehistoric coast. Another well-known pterosaur, the late Jurassic Rhamphorhynchus, had a long, curved, pointed snout and prominent teeth to match. This species is known from Europe, and lived during a time when this area was a hot, dry archipelago in a shallow, salty sea. Thanks to this, Rhamphorhynchus is known from several exquisitely preserved specimens, some of which preserve impressions of the wing and tail membranes as well as gut contents. Rhamphorhynchus preyed on sea life like fish and cephalopods which it may have caught in its toothy jaws while swimming. This pterosaur may have fallen prey to other sea creatures itself, as one specimen was found caught in the jaws of the gar-like fish Leptolepides (Frey & Tischlinger, 2012). The small anurognathans, which could be described as ‘fuzzy, winged frogs’ may have lived something like modern bats or nighthawks. Their big eyes and broad mouths with needle-like teeth appear useful for hunting insects in low lighting.
In the more derived pterodactyloids, some lineages get truly strange. Close cousins to Pterodactylus itself, the ctenochasmatids produced some species with long snouts full of thin, needle-like teeth. Perhaps the most bizarre of these was Pterodaustro, which had an elongated, recurved snout and a lower jaw filled with about a thousand long, bristle-like teeth. It’s thought that this pterosaur might have waded through shallow water, straining small invertebrates with its teeth which acted like a filter. This lifestyle is now taken up by birds like some ducks, geese, and flamingos who utilize keratinous bristles in their bills as opposed to teeth.
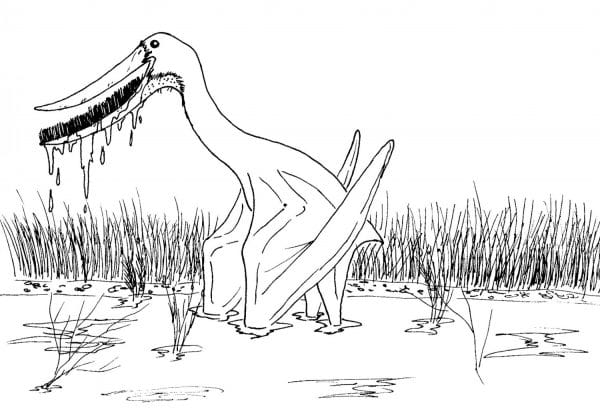
Pterodaustro strains aquatic invertebrates out of a wetland with its countless teeth. By Nicholas Carter
Moving up the pterosaur tree, we encounter the pteranodonts, containing perhaps the most recognizable pterosaur of all, Pteranodon. With its backwards-facing crest and long, curved beak, Pteranodon has been featured in popular depictions of pterosaurs for a hundred years. Discovered from the Kansas chalk dating back to mid-late Cretaceous, Pteranodon lived in the skies over the Western Interior Seaway, and its lifestyle is often compared to modern large oceanic birds like albatrosses and pelicans. Two forms of Pteranodon are known- a larger one with a big, rear-projecting crest and a narrow pelvis, and a smaller one with a little, nub-like crest and wider pelvis. These forms are sometimes interpreted as male and female respectively, with the males having larger display crests and females a wider pelvis (to accommodate eggs) (Bennett, 1992). Pteranodon’s diet was mostly fish-based, and it might have swam or even dove like a gannet after its prey, or maybe nabbed it during flight. There’s nothing to suggest, though, that pterosaurs, including Pteranodon, were limited to the most obvious prey that they seemed adapted to. Looking at an example in birds, you don’t have to look hard to find footage of pelicans, which survive mostly on fish, snapping up and swallowing entire smaller birds alive and kicking. Animals don’t care what we think they should be doing, they’ll do what they want. A related species to Pteranodon, Nyctosaurus, was highly modified with its massive fork-shaped crest. It also completely lost its hand digits aside from the wing finger, puzzlingly enough, and may have spent very little time on solid land (Bennett, 2003). Further along the pteranodont lineage are the toothed ornithocheiromorphs, including large carnivores like Tropeognathus and Anhanguera which bore large, round crests on the tops and bottom jaws near the end of the snout.
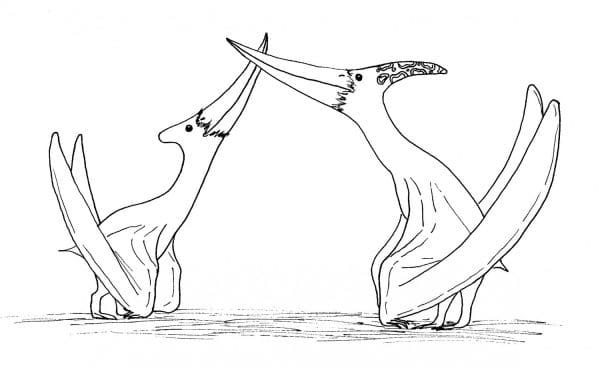
Male and female Pteranodon engaging in a courtship ritual. Many animals perform behaviours like this at certain times of the year, and perhaps pterosaurs did too. By Nicholas Carter
Sister group to the pteranodonts were the azhdarchoids. This included groups like the tapejarids, a showy lineage with elaborate crests and toothless beaks which may have lived in trees, eating fruits and seeds like prehistoric parrots (Wu et al., 2017). Another strange group was the dsungaripterids. The namesake of this family, Dsungaripterus, bore an unusual triangle-shaped skull topped with a tiny crest. The tip of its beak was pointed and upturned, and the back of it jaws bore flat, crushing teeth. This pterosaur may have lived somewhat like a shorebird, overturning rocks and probing mud with its beak and crushing shelled invertebrates with its teeth.
The kings of the pterosaur world were the azhdarchid family. While some members of this group weren’t terribly impressive, some species could grow to enormous sizes, as tall as giraffes or bigger. Species like this include the famous Quetzalcoatlus of North America, Hatzegopteryx of Romania, and Arambourgiania of Jordan. These creatures were oddly built even amongst pterosaurs, with the skull and neck alone often measuring half the body length if not more. It’s hard to imagine such front-loaded animals flying, and some have argued that these big azhdarchids were secondarily flightless, but this idea has not received much support (Witton et al., 2010). It’s tempting to imagine the giant azhdarchids swooping down over the ocean to skim prey from the surface, or descending over the landscape to snatch up unwary dinosaurs and other animals, but current research suggests they actually hunted on the ground as giant terrestrial stalkers (Witton & Naish, 2008). Quetzalcoatlus existed alongside Tyrannosaurus in western North America at the end of the Cretaceous. It’s hard to think of a more dangerous ecosystem.
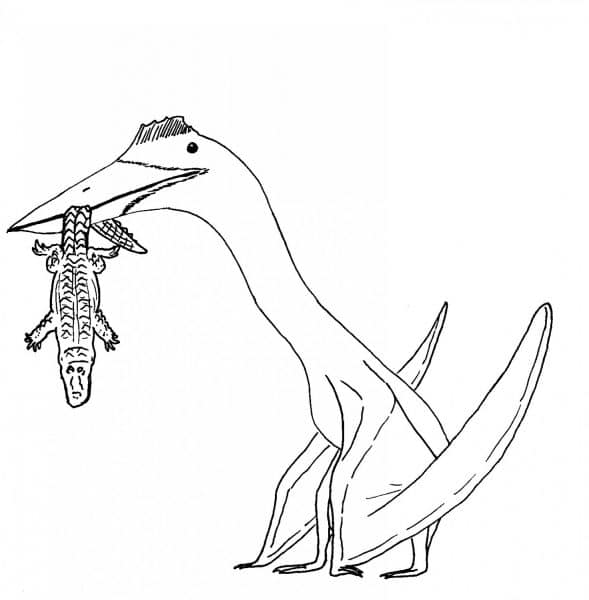
Big azhdarchids likely hunted on the ground, snatching up prey in their big toothless beaks like storks do today. Big azhdarchids were all head and neck. By Nicholas Carter
Big azhdarchids are known from Alberta, but from relatively fragmentary remains. Considering the fragility of pterosaur bones and the bias towards preservation of big, sturdy animals in the rocks here, our pterosaur remains are a precious few. One particular specimen, an azhdarchid tibia from Dinosaur Provincial Park, was found with a dromaeosaur tooth embedded in it- perhaps an instance of scavenging. (Godfrey & Currie, 2005). Pterosaur vertebrae and wing bones have also been found in this area. Nearer to the Currie Museum in the Wapiti Formation, no pterosaur remains have been found yet. However, in 2012 paleontologists with the Boreal Alberta Dinosaur Project reported a possible large pterosaur hand print from west just of Grande Prairie (Bell et al., 2012). There’s so sure way to know what exactly the track maker was, but it may have been some kind of large Quetzalcoatlus-like creature. Given the distance big pterosaurs like this could have travelled with relative ease, they were certainly present here millions of years ago.
This is just a glance at the tip of the iceberg that is pterosaur biology and evolution. It’s a shame that these fascinating creatures are so often stuck in the shadows of the dinosaurs, despite flying high above them.
By Nicholas Carter
References
Bell, P.R., Fanti, F. & Sissons, R. 2012: A possible pterosaur manus track from the Late Cretaceous of Alberta. Lethaia, DOI: 10.1111/let.12006
Bennett, S.C. (1992). “Sexual dimorphism of Pteranodon and other pterosaurs, with comments on cranial crests”. Journal of Vertebrate Paleontology. 12(4): 422–434.
Bennett SC (2000). “Pterosaur flight: the role of actinofibrils in wing function”. Historical Biology. 14(4): 255–84.
Bennett, S.C. (2003). “New crested specimens of the Late Cretaceous pterosaur Nyctosaurus.” Paläontologische Zeitschrift, 77: 61-75.
Benton, M. J. (1999). “Scleromochlus taylori and the origin of dinosaurs and pterosaurs”. Philosophical Transactions of the Royal Society B: Biological Sciences. 354 (1388): 1423–1446. doi:10.1098/rstb.1999.0489. JSTOR 57034. PMC 1692658.
Benton, Michael J.; Xu, Xing; Orr, Patrick J.; Kaye, Thomas G.; Pittman, Michael; Kearns, Stuart L.; McNamara, Maria E.; Jiang, Baoyu; Yang, Zixiao (January 2019). “Pterosaur integumentary structures with complex feather-like branching”. Nature Ecology & Evolution. 3 (1): 24–30.
Frey E, Tischlinger H (2012) The Late Jurassic Pterosaur Rhamphorhynchus, a Frequent Victim of the Ganoid Fish Aspidorhynchus? PLoS ONE 7(3): e31945. https://doi.org/10.1371/journal.pone.0031945
Godfrey, S.J., and Currie, P.J. 2005. Pterosaurs. In: Currie, P.J., and Koppelhus, E.B. (eds), Dinosaur Provincial Park: A Spectacular Ancient Ecosystem Revealed. Indiana University Press: Bloomington and Indianapolis, p. 292-311.
Unwin DM (1997). “Pterosaur tracks and the terrestrial ability of pterosaurs”. Lethaia. 29 (4): 373–86.
Witton MP, Naish D (2008) A Reappraisal of Azhdarchid Pterosaur Functional Morphology and Paleoecology. PLoS ONE 3(5): e2271. https://doi.org/10.1371/journal.pone.0002271
Witton, Mark P.; Habib, Michael B.; Laudet, Vincent (15 November 2010). “On the Size and Flight Diversity of Giant Pterosaurs, the Use of Birds as Pterosaur Analogues and Comments on Pterosaur Flightlessness”. PLoS ONE. 5 (11): e13982. doi:10.1371/journal.pone.0013982.
Wu, Wen-Hao; Zhou, Chang-Fu; Andres, Brian (2017). “The toothless pterosaur Jidapterus edentus (Pterodactyloidea: Azhdarchoidea) from the Early Cretaceous Jehol Biota and its paleoecological implications”. PLoS ONE. 12 (9): e0185486. doi:10.1371/journal.pone.0185486.